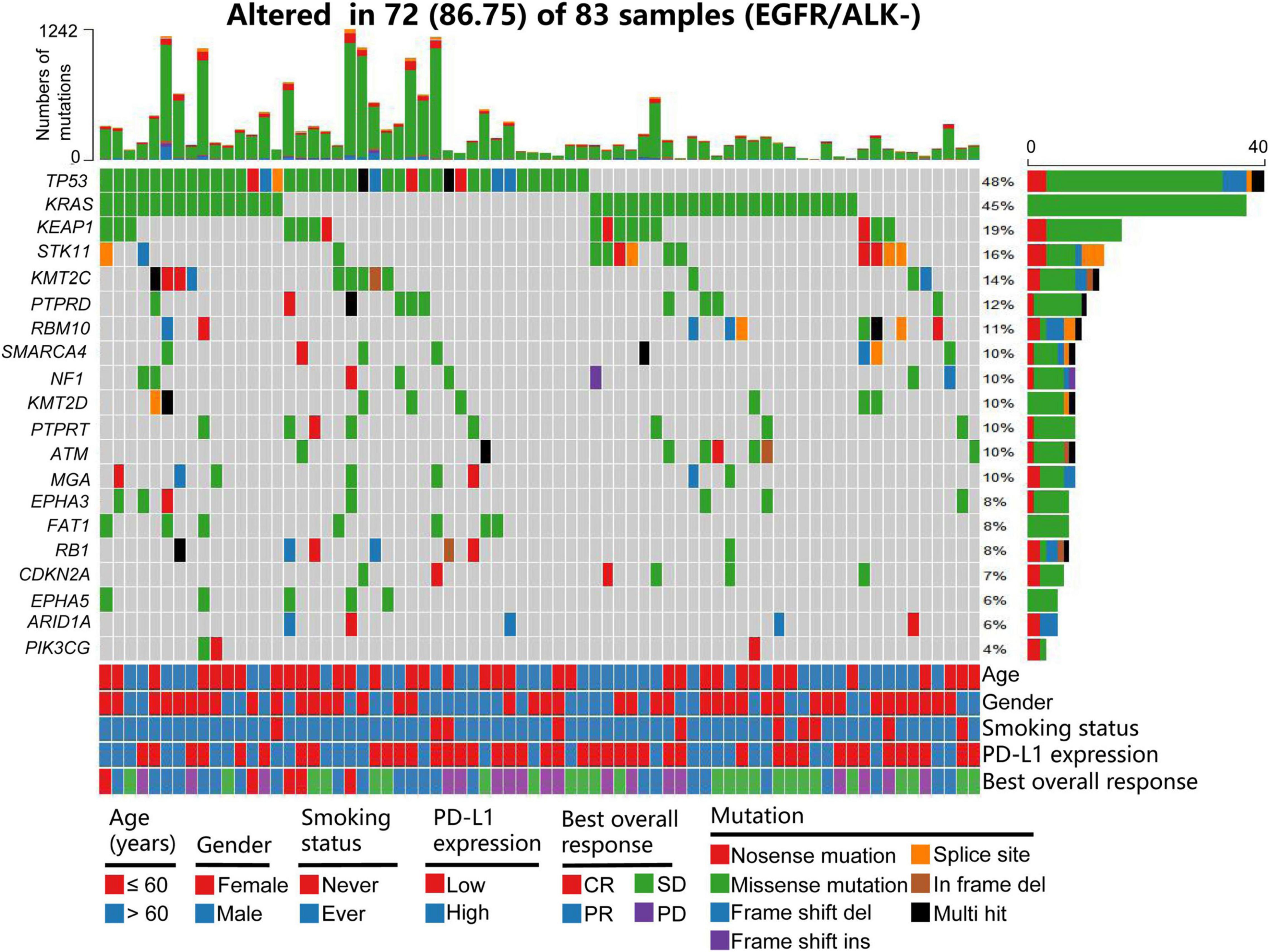
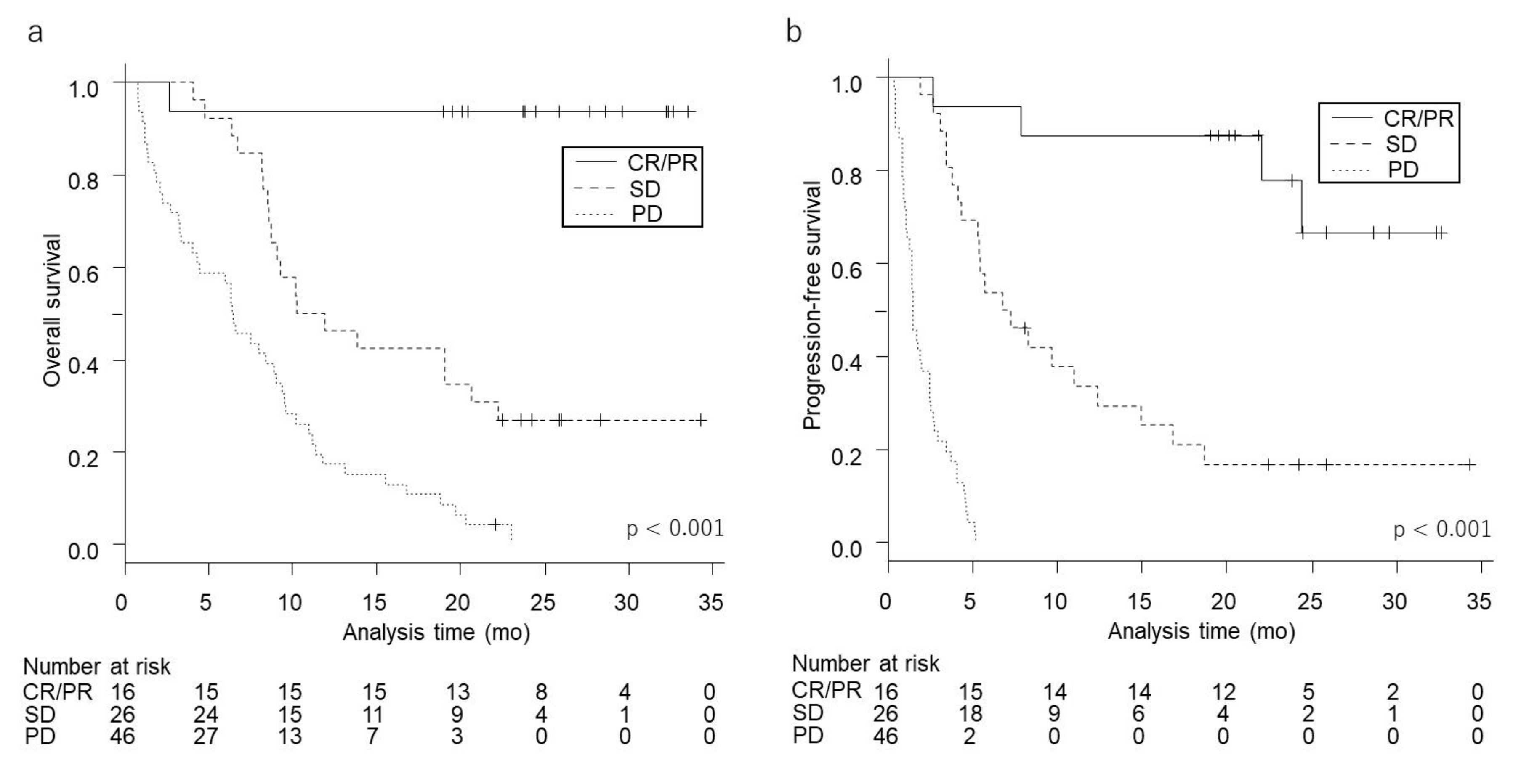
Frontiers | A Somatic Mutation Signature Predicts the Best Overall Response to Anti-programmed Cell Death Protein-1 Treatment in Epidermal Growth Factor Receptor/Anaplastic Lymphoma Kinase-Negative Non-squamous Non-small Cell Lung Cancer

Table 3 from New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). | Semantic Scholar
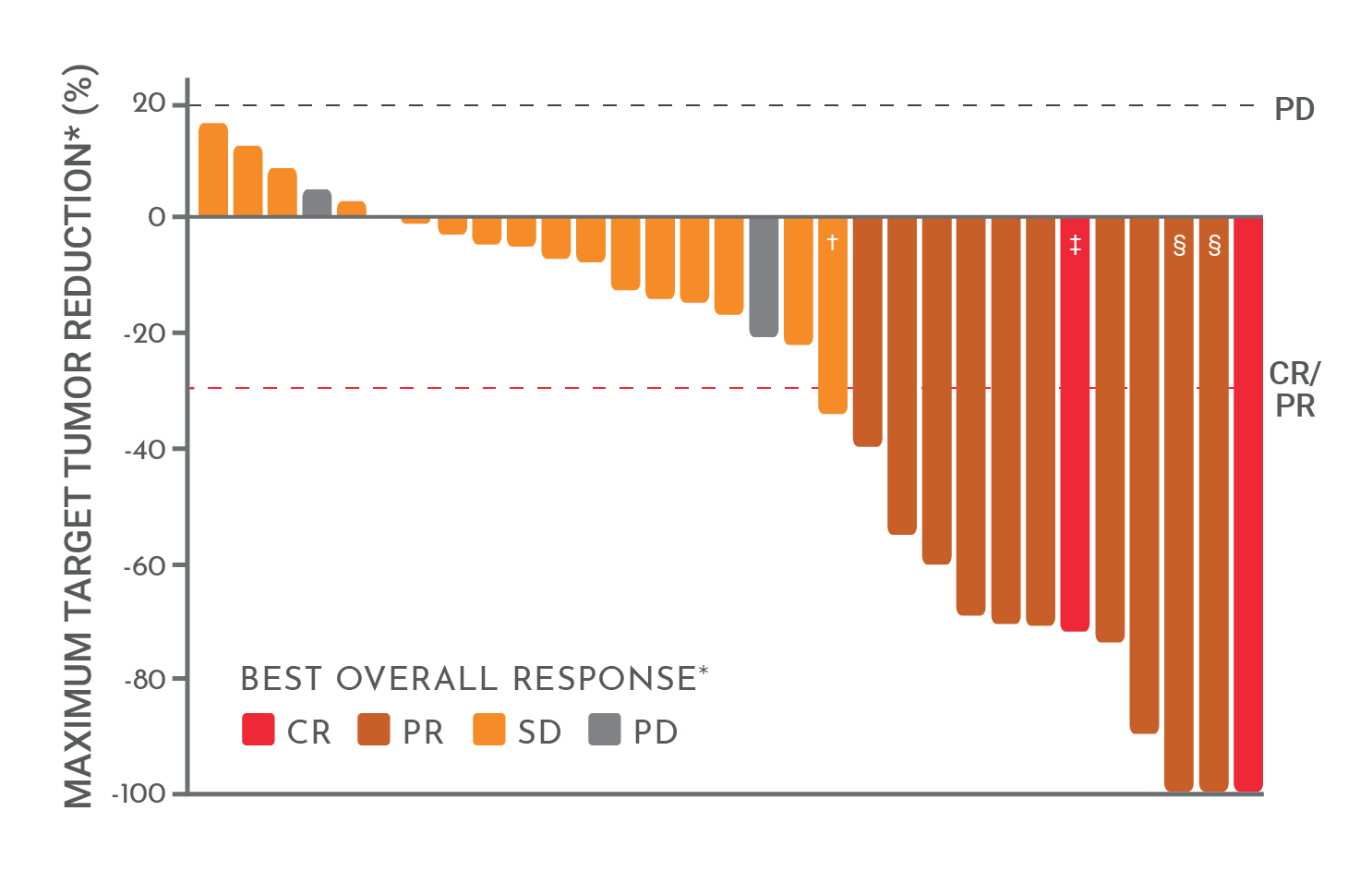
Cancers | Free Full-Text | Real-World, Long-Term Outcomes of Nivolumab Therapy for Recurrent or Metastatic Squamous Cell Carcinoma of the Head and Neck and Impact of the Magnitude of Best Overall Response:

Best overall response (per RECIST 1.1, based on investigator assessment) | Download Scientific Diagram
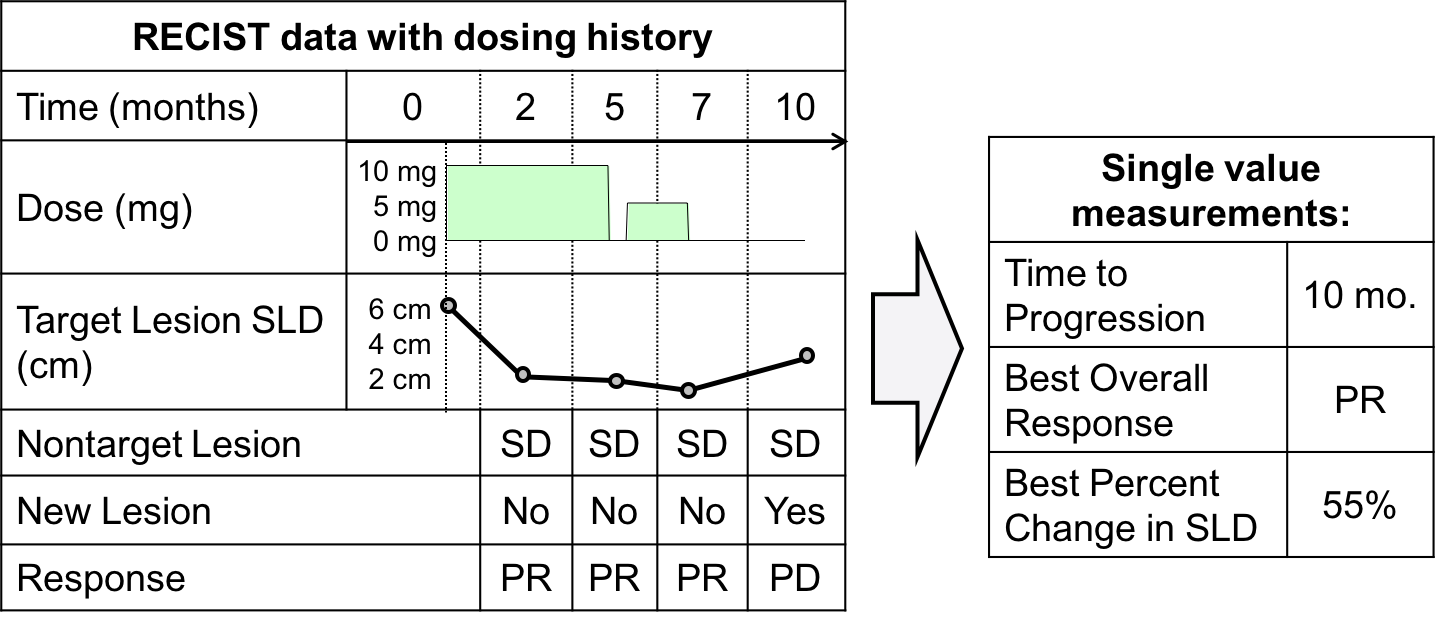
Simplifying the Derivation of Best Overall Response per RECIST 1.1 and iRECIST in Solid Tumor Clinical Studies

Explicyte and Institut Bergonié associate in a clinical trial report publication - by Explicyte Immuno-Oncology

Response evaluation for immunotherapy through semi-automatic software based on RECIST 1.1, irRC, and iRECIST criteria: comparison with subjective assessment - Ying-Chieh Lai, Wen-Cheng Chang, Chun-Bing Chen, Chi-Liang Wang, Yu-Fen Lin, Ming-Mo Ho,

Andy Biotech on Twitter: "$MRTX very misleading and pathetic to include unconfirmed responses that will not be confirmed in ORR% RECIST guideline clearly requires "in non-randomised trials where response is the primary
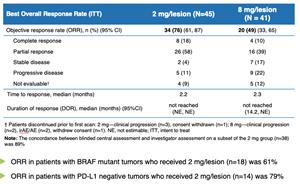
Overall Response Rate of 76% in Advanced Melanoma Patients with Dynavax's SD-101 in Combination with KEYTRUDA® (pembrolizumab); Data Presented Today at the 2019 ASCO Annual Meeting | Dynavax Technologies Corporation







![PDF] Efficacy endpoints in Oncology | Semantic Scholar PDF] Efficacy endpoints in Oncology | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/aeb34663ace6e18c7e6a588ae474e466bb3e514b/2-Table1-1.png)